Похожие презентации:
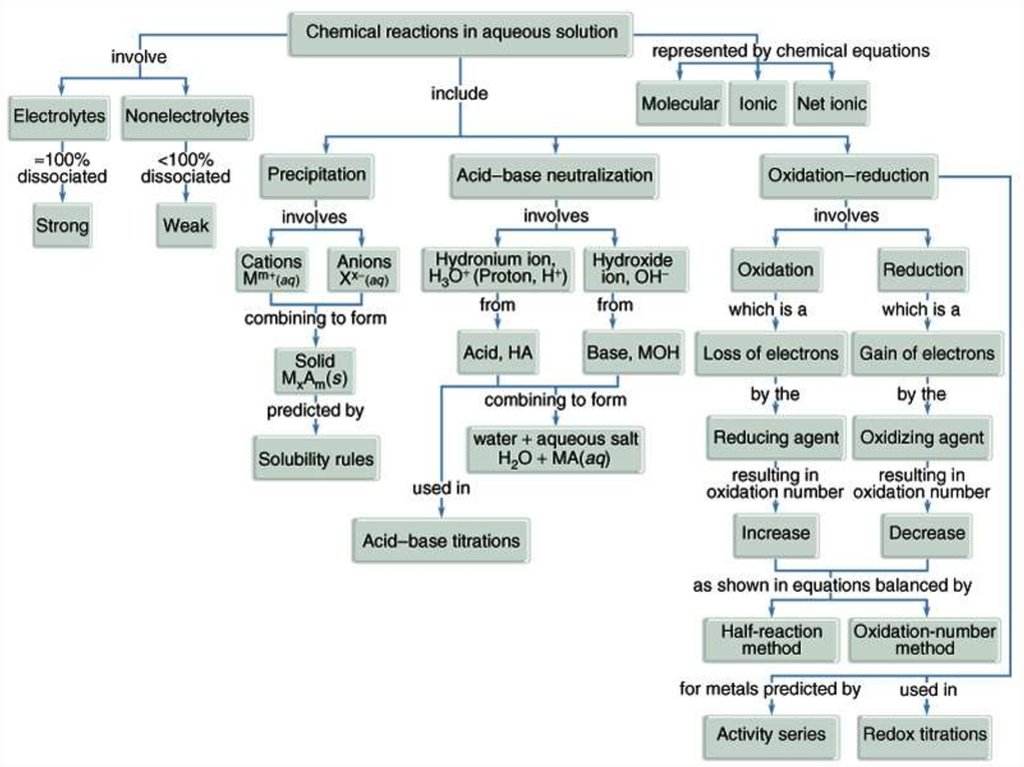
Oxidation – Reduction Reaction
1.
LECTURE № 10Oxidation – Reduction
Reaction
07.04.2017
2.
3.
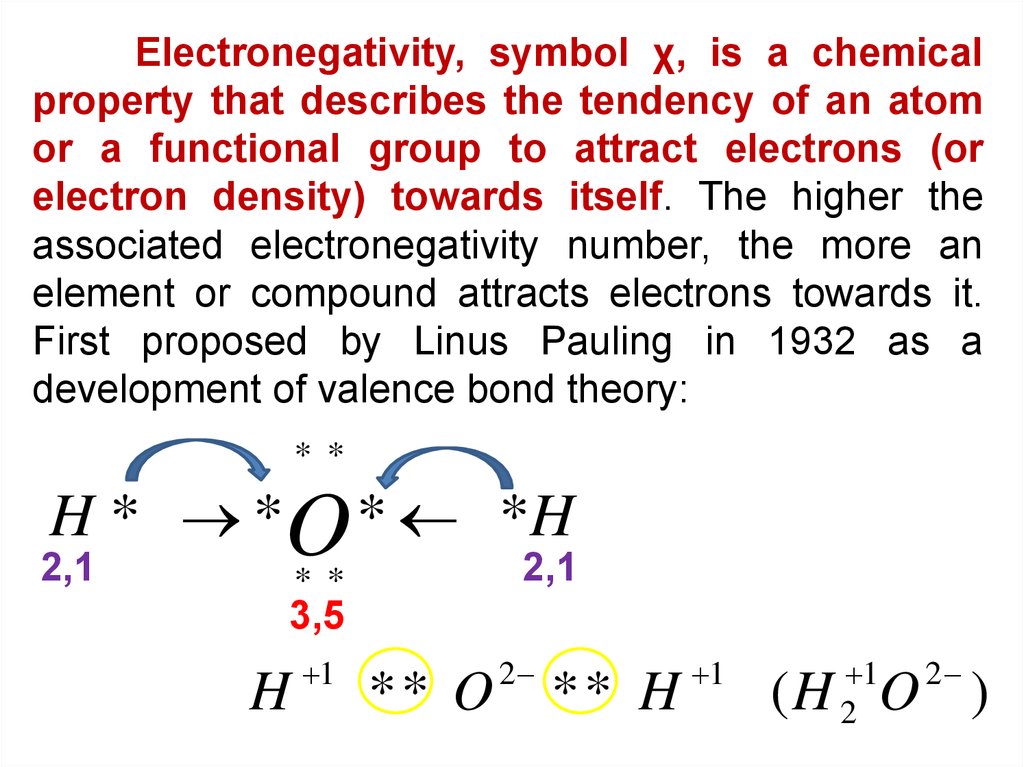
Electronegativity, symbol χ, is a chemicalproperty that describes the tendency of an atom
or a functional group to attract electrons (or
electron density) towards itself. The higher the
associated electronegativity number, the more an
element or compound attracts electrons towards it.
First proposed by Linus Pauling in 1932 as a
development of valence bond theory:
* *
H * *O * *H
2,1
2,1
* *
3,5
H
1
** O
2
** H
1
1
2
2
(H O )
4.
ЭЛЕКТРООТРИЦАТЕЛЬНОСТЬ ЭЛЕМЕНТОВэто способность одних атомов оттягивать на себя электроны от
других атомов при взаимодействии
5.
Not all atoms attract electrons with the same force. Theamount of "pull" an atom exerts on its electrons is called its
electronegativity. Atoms with high electronegativities — such
as fluorine (4,0), oxygen (3,5), and nitrogen (3,0) — exert a
greater pull on electrons than atoms (metals have <1,5) with
lower electronegativities. In a bond, this leads to unequal sharing
of electrons between the atoms, as electrons will be drawn closer to
the atom with the higher electronegativity.
Because electrons have a negative charge, the unequal
sharing of electrons within a bond leads to the formation of an
electric dipole: a separation of positive and negative electric charge.
Because the amount of charge separated in such dipoles is usually
smaller than a fundamental charge, they are called partial charges,
denoted as δ+ (delta plus) and δ− (delta minus). These symbols
were introduced by Christopher Ingold and his wife Hilda Usherwood
in 1926. The bond dipole moment is calculated by multiplying the
amount of charge separated and the distance between the charges.
These dipoles within molecules can interact with dipoles in
other molecules, creating dipole-dipole intermolecular forces.
6.
An amount of electrons which was lost orgained by the atom in a chemical compound is
called the oxidation state, often called the
oxidation number. Oxidation states are
typically represented by small integers.
Conceptually, the oxidation state, which
may be positive (cations Na+1, Mg2+, Al3+),
negative (O2-, Cl-, NO3-, S2-, SO42-, PO43-) or zero
(free atoms Na , H2 , O2 , P ), is the
hypothetical charge that an atom would have if
all bonds to atoms of different elements were
100% ionic, with no covalent component. This is
never exactly true for real bonds.
7.
Rules:Examples
Exceptions:
Free elements (uncombined state)
and pure compounds have an
oxidation number of zero.
Na , H2 , O2 , P
CaCl2 , H2O , H3PO4
Fluorine always has a -1 oxidation
number within compounds
F2-1O, HF-1, CaF2-1
The oxidation number of oxygen in
compound is usually –2
CO22-, Na2O2-
H2O2-1, F2O2+
The oxidation number of hydrogen in
compound is usually +1
H+1Cl, H2+1O, NH3+1,
CH4+1
metal hydrides:
NaH-1, CaH2-1, AlH3-1
Alkali metal atoms (Group I) have an
Li+, Na+, K+, Rb+, Cs+
oxidation number equal to +1 within
compounds. Alkali earth atoms (Group Mg2+ , Ca+2 , Ba+2, Sr+2
II) have an oxidation number of +2
within compounds.
The algebraic sum of oxidation states
of all atoms in a neutral molecule
must be zero
[K2+1Cr2+6O7-2]
While in ions the algebraic sum of the
oxidation states of the constituent
atoms must be equal to the charge on
the ion
[P+5O4-2]3-
2*(+1)+2*(+6)+7*(-2)=0
1*(+5)+4*(-2)=-3
8.
The increase in oxidation state of anatom through a chemical reaction is known
as an oxidation (окисление):
Fe 2 e Fe
2
a decrease in oxidation state is known as a
reduction (восстановление):
O 2 e 2 2O
2
2
4e
2
2
2 Fe O 2[ Fe O ]
2
9.
In 1streaction no change of oxidation
degrees of atoms:
1
2
Na O H
1
1
H Cl
1
1
Na Cl
1
1
2
H O
2
In 2nd equation we see that Mn and N atoms
change their oxidation states:
1
7
K Mn O
2
4
1
3
Na N O
2
2
1
2
6
H S O
2
4
Mn 2 S 6 O4 2 K 2 1 S 6 O42 Na 1 N 5 O32 H 2 1O 2
10.
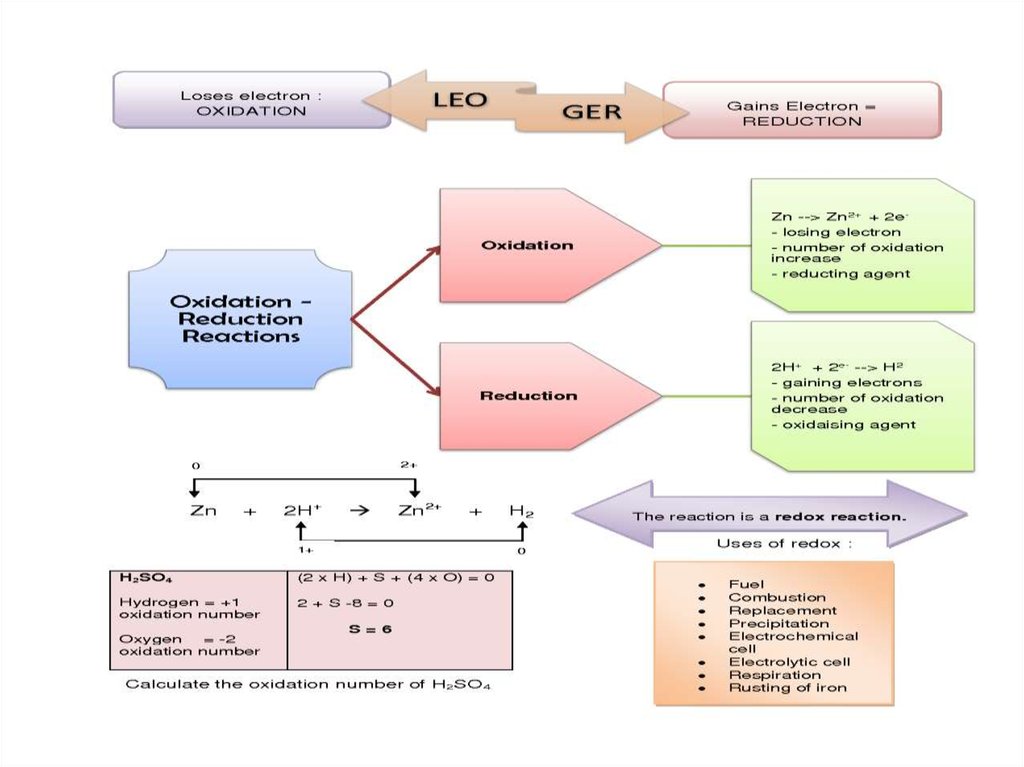
Oxidation-Reduction Reactions are allreactions that involve the change of an
oxidation number, and transfer of electrons
among the reacting substances.
• Oxidation is the loss of electrons or
an
increase
in
oxidation
state
by
a molecule, atom, or ion.
• Reduction is the gain of electrons or
a decrease in oxidation state by a molecule,
atom, or ion.
11.
The chemical substance which loseselectrons, is oxidised (as result increases
its valency) and called the reducing agent
(reductant).
The chemical substance which gains
electrons is reduced (as result decreases
its valency) and called the oxidising agent
(oxidant).
2 Fe O 2 FeO
reducing
agent
2
oxidizing
agent
12.
IMPOTANT OXIDANTSAn oxidizing agent: contains an element whose
oxidation state decreases in a redox reaction gains
electrons:
1) Halogens in free state: F2 Cl2 I2 Br2
2) Oxigen O2 and ozon O3
3) Nitrogen N2
4) Oxygen-containing mineral acids: nitric acid
(HNO3), perchloric acid (HClO4), and sulfuric acid
(H2SO4)
5) Permanganate (MnO4-), chromate (CrO42-), and
dichromate (Cr2O72-) ions in acidic solution
6) Oxides: CuO, PbO2, Ag2O
7) Compounds of Iron (III) ion (Fe3+): FeCl3
13.
IMPOTANT REDUCTANTSA reducing agent: contains an element whose
oxidation state increases in a redox reation loses
electrons:
1) Alkali metal atoms (Group I) and Alkali earth metal
atoms (Group II): Na, K, Mg,Ca
2) Average activity metals: Zn, Al, Fe
3) Hydrogen H2
4) Hydrides of alkali and alkaline earth metals: NaH, CaH2
5) Some non-metals: P, Si, C (coal)
6) Hydrohalogen acids and their salts: HCl, HI, HBr
7) Compounds of Iron (II) ion (Fe2+): FeSO4 and FeCl2
8) Metal cations in the lower oxidation state: Sn2+ Sn4+
9) Nitrous acid HNO2, ammonia NH3
10) H2S, CO, SO2
14.
Some substances containing elementsin the intermediate oxidation may be
oxidants or reductants:
1) Halogens in basic solution (exception only
F 2)
- 6e
0
-1
+5
I 2 NaOH NaI NaIO3 H 2 O
+2e
2) Hydrogen peroxide H2O2
3) Sulfurous acid and its salt H2SO3
15.
oxidantH2+O–2
2О– +2е 2О-2
О20
2О– -2е О20
H2+O2–
reductant
Na2S–2
S+4 +6e S–2
oxidant
Na2S+4O3
+4e
S0
S+4 +4e S0
Na2S+6O4
S+4 – 2e S+6
reductant
16.
REDOX SCALEReducing agents are oxidised. Oxidation is the loss of electrons (-ne)
+6е
-2е
0
-6
-5
-4
-3
-2
-1
+1
+2
+3
+4
+5
+6
+7
Oxidising agents are reduced. Reduction is the gain of electrons (+ne)
+1 +5 -2
-2е
+1 -1
0
KClO3 KCl O2
+6е
17.
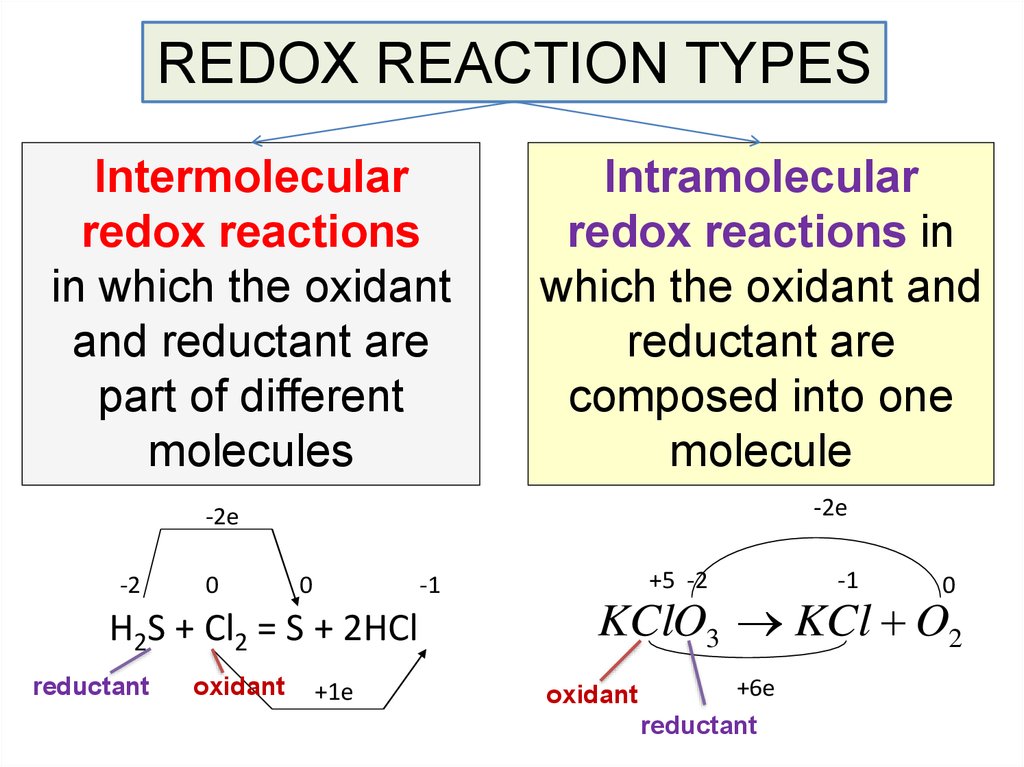
REDOX REACTION TYPESIntermolecular
redox reactions
in which the oxidant
and reductant are
part of different
molecules
Intramolecular
redox reactions in
which the oxidant and
reductant are
composed into one
molecule
-2е
-2е
-2
0
0
-1
H2S + Cl2 = S + 2HCl
reductant
oxidant
+1е
+5 -2
-1
0
KClO3 KCl O2
oxidant
+6е
reductant
18.
DISPROPORTIONATION REACTIONSIn
some
redox
reactions,
called
disproportionation reactions, the same substance is
both oxidized and reduced.
Disproportionation
reaction
is
a
intramolecular reaction in which the atoms of one
element is reduced and simultaneously increase
the degree of oxidation state.
+2e
ЗСl20 + 6КОН = 5КСl-1 + КСl+5О3 + ЗН2О
oxidant
reductant
- 5e
19.
DISPROPORTIONATION REACTIONS-2е
+5
+7
-1
4KClO3 3KClO4 KCl
+6е
+1е
-1
0
+1
Br2 H 2O HBr HBrO
-1е
20.
METHODS OF BALANCING REDOXREACTIONS
1) The Oxidation-Number Method (ionic
balance): the key to this method is to realize that the net
change in the total of all oxidation numbers must be zero.
That is, any increase in oxidation number for the oxidized
atoms must be match by the corresponding decrease in
oxidation number for the reduced atoms.
Cu HNO3 Cu ( NO3 ) 2 NO2 H 2 O
Cu H 1 NO3 1 Cu 2 2 NO3 1 NO2 H 2 O
Cu 2e Cu 2
2
1
oxidation, reductant
NO3 1 2 H 1 1e NO2 H 2 O
1
2
reduction, oxidant
-1+2(+1)=+1
0+0=0
21.
2) Halfbalance):
Reactions
Method
(electronic
1) Write and balance separate half-equations for oxidation and
reduction, and balance the equation.
2) Adjust coefficients in the two half-equation so that the same
number of electrons appears in each half equation.
3) Add together the two half-equation, then cancel the species
common to both side of the equation to obtain the balanced
overall equation.
1
5
2
3
K Cl O
Cl
5
2O
2
1
K Cl
6e Cl
1
2e 2 O
2
1
O
2
6
4=2
reduction, oxidant
4
6=3
oxidation, reductant
2 KClO3 2 KCl 3O2
22.
H+ (+5e)+7
KMnO4
permanganate ion
(purpule sln)
H2O (+3e)
OH– (+1e)
Mn+2
colorless
+4
MnO2
+6
K2MnO4
yellow
green
In acidic solution:
2KMnO4 + 5Na2SO3+ 3H2SO4 = 2MnSO4+ 5Na2SO4 + K2SO4 + 3H2O
In neutral aqueous solution:
2 KMnO4 + 5Na2SO3+ H2O = 2MnO2+ 3 Na2SO4 + 2KOH
In basic solution:
2 KMnO4 + Na2SO3+ 2КOH = 2 K2MnO4+ Na2SO4 + H2O
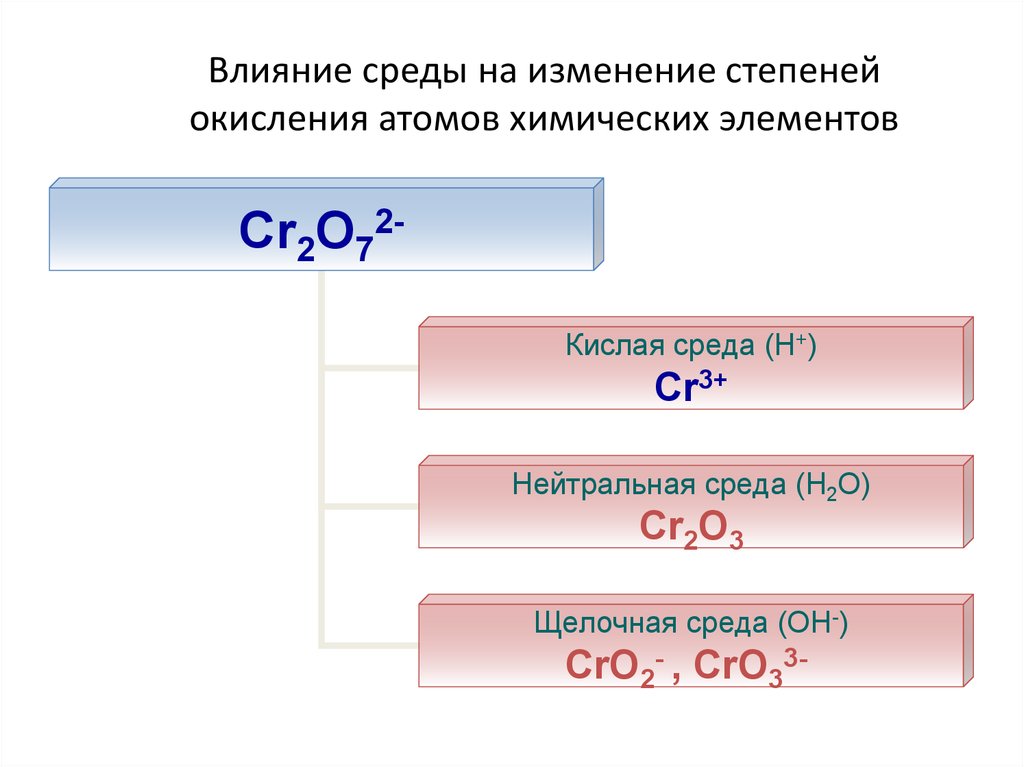
23. Влияние среды на изменение степеней окисления атомов химических элементов
Cr2O72Кислая среда (Н+)Cr3+
Нейтральная среда (Н2О)
Cr2O3
Щелочная среда (ОН-)
CrO2- , CrO33-
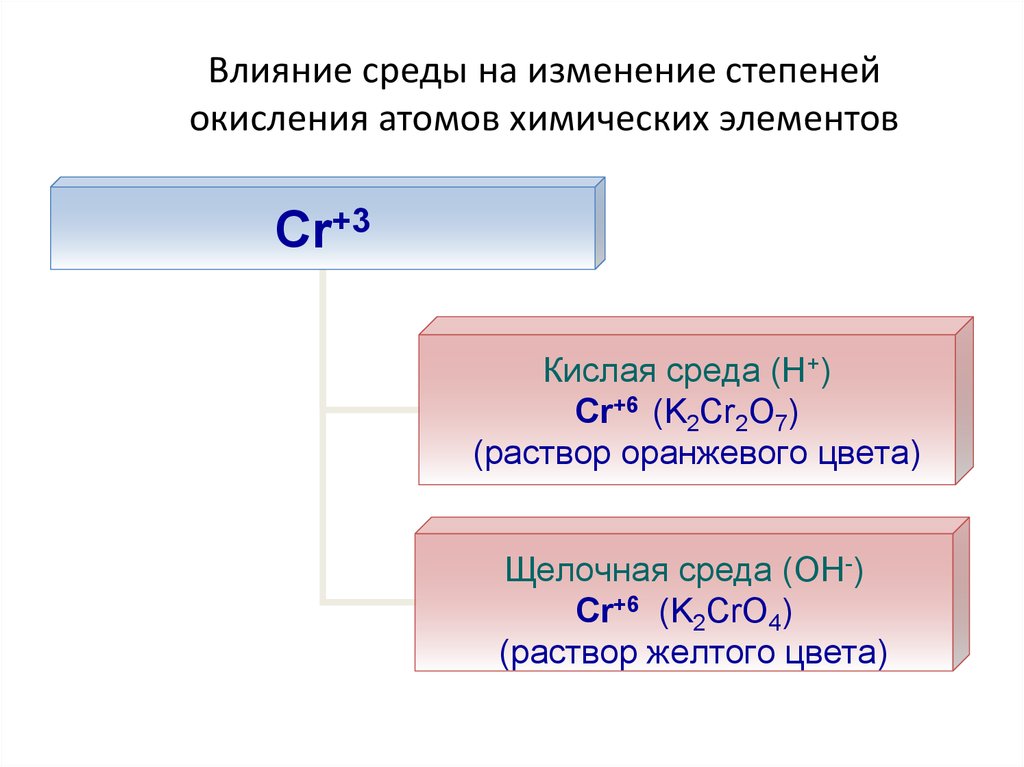
24. Влияние среды на изменение степеней окисления атомов химических элементов
Cr+3Кислая среда (Н+)
Cr+6 (K2Cr2O7)
(раствор оранжевого цвета)
Щелочная среда (ОН-)
Cr+6 (K2CrO4)
(раствор желтого цвета)
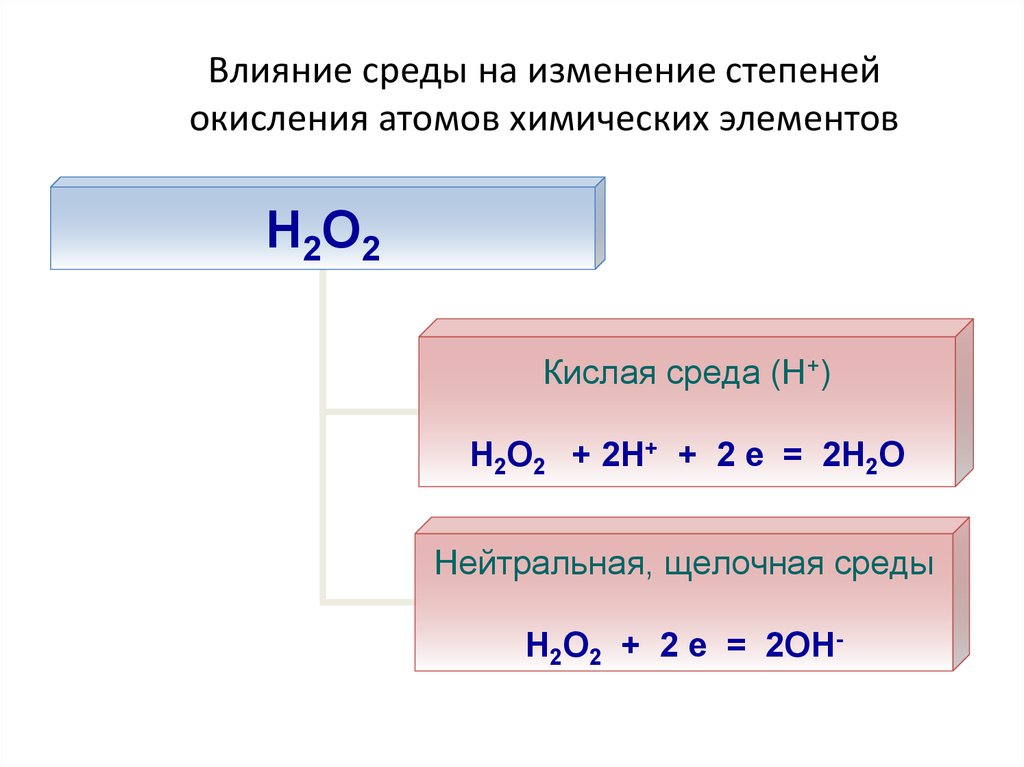
25. Влияние среды на изменение степеней окисления атомов химических элементов
Н2О2Кислая среда (Н+)
Н2О2 + 2Н+ + 2 е = 2Н2О
Нейтральная, щелочная среды
Н2О2 + 2 е = 2ОН-
26.
GLOSSARYOxidation Any chemical change in which at least one element loses electrons, either
completely or partially.
Reduction Any chemical change in which at least one element gains electrons, either
completely or partially.
Oxidation-reduction reactions The chemical reactions in which there is a complete or
partial transfer of electrons, resulting in oxidation and reduction. These reactions are also
called redox reactions.
Half-reactions Separate oxidation and reduction reaction equations in which electrons
are shown as a reactant or product.
Reducing agent A substance that loses electrons, making it possible for another
substance to gain electrons and be reduced.
Oxidizing agent A substance that gains electrons, making it possible for another
substance to lose electrons and be oxidized.
Oxidation number A tool for keeping track of the flow of electrons in redox reactions
(also called oxidation state).
Combination or synthesis reaction The joining of two or more elements or compounds
into one product.
Decomposition reaction The conversion of one compound into two or more simpler
substances.
Combustion reaction Rapid oxidation accompanied by heat and usually light.
Single-displacement reaction Chemical change in which atoms of one element displace
(or replace) atoms of another element in a compound.




















 Химия
Химия








